What is the difference between the endpoint equivalence point and indicator?
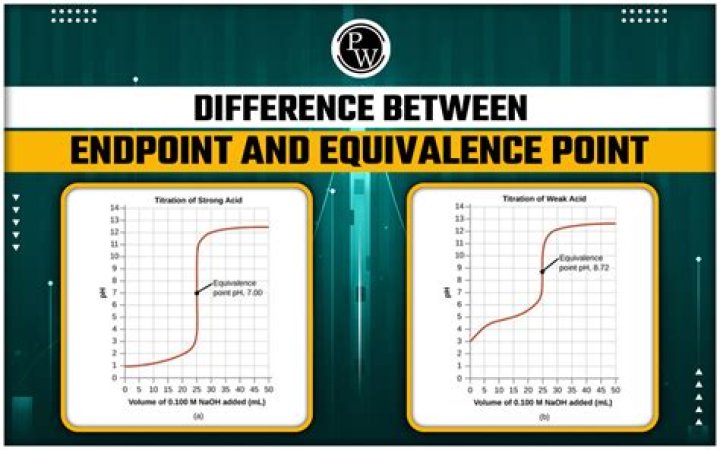
The main difference between an equivalence point and an endpoint is that the former marks the end of the reaction whereas the latter is a point where the indicator changes colour….Endpoint vs Equivalence Point.
| Endpoint | Equivalence Point |
|---|---|
| Comes after the equivalence point | Comes before the endpoint |
What is the indicator endpoint?
The point at which the indicator changes color is called the endpoint. So the addition of an indicator to the analyte solution helps us to visually spot the equivalence point in an acid-base titration. Endpoint: refers to the point at which the indicator changes color in an acid-base titration.
Do indicators show equivalence point?
A pH indicator shows the equivalence point —the point at which the equivalent number of moles of a base have been added to an acid. It is often wrongly assumed that neutralization should result in a solution with pH 7.0; this is only the case in a strong acid and strong base titration.
Why is it important to match the endpoint of a pH indicator to the equivalence point of the titration?
This rapid rise causes the indicator to change color, so the endpoint matches the equivalence point if the indicator is chosen properly. Titration of a strong base with a strong acid can be handled in essentially the same way as the strong acid-strong base situation we have just described.
Is equivalence point the same as equilibrium?
At the equivalence point, the amount of base and acid are exactly equal. Titration of a weak acid with a strong base has the same four regions. However there is an equilibrium associated with such a problem.
What is the equivalence point?
The equivalence point or stoichiometric point is the point in a chemical reaction when there is exactly enough acid and base to neutralize the solution. In a titration, it is where the moles of titrant equal the moles of solution of unknown concentration.
How do you find the equivalence point?
On the curve, the equivalence point is located where the graph is most steep. There is a fast and abrupt change of pH around this point, which can be observed by the color change the takes place during titration. At the equivalence point, an ICE table is required to determine volume and acidity.
Why does endpoint come after equivalence point?
Equivalence point means that the titrant has reacted fully with the analyte whereas the endpoint signals the completion of titration. Endpoint and equivalence can occur at the same time if the pH of the titrant corresponds to the pH at equivalence point.
Why does pH jump at equivalence point?
Near the equivalence point, a change of a factor of 10 occurs very quickly, which is why the graph is extremely steep at this point. As the hydronium ion concentration becomes very low, it will again take a lot of base to increase the hydroxide ion concentration by 10 fold to change the pH significantly.
What is the equivalence point of indicator?
Indicators can be used for this purpose, for example, methyl orange or phenolphthalein. The equivalence point cannot be taken the same as the endpoint of a titration. The endpoint is referred to as the point at which the used indicator changes its color. These color changes appear after reaching the equivalence point.
What is the difference between equivalence point and end point?
The end of the reaction is given by the equivalence point. The endpoint indicates that the equivalence point has been reached. The main difference between equivalence point and endpoint is that equivalence point is the actual point where the chemical reaction ends whereas end point is the point where the color change occurs in the system.
What is equivalence point and endpoint in titration?
The point in the titration process where the chemical reaction in the titration mixture ends is called equivalence point. The point in the titration process which is indicated by color change of the indicator is called endpoint. It is the point where the analyte has completely reacted with the titrant.
What is the endpoint and equivalence point of half reaction?
Therefore, the endpoint is equal to the equivalence point here. For example, the half-reaction of conversion of Cr 2 O 72- to C 3+ changes the color of the medium from orange to green. This is because the color of Cr (+6) in aqueous solution is orange and the color of Cr (+3) in the aqueous solution is green.