What is refining of copper?
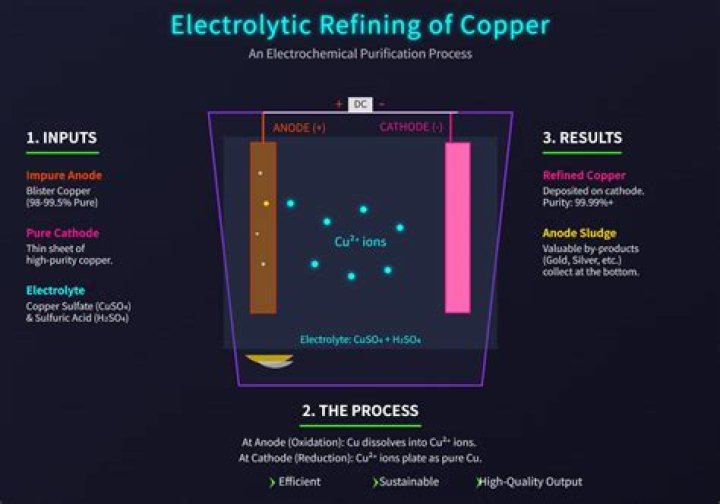
Electrolytic refining is a process of refining a metal (mainly copper) by the process of electrolysis. The clean or pure metal is formed at the cathode when the electrical current of a sufficient voltage is applied by dissolving impure metal at the anode.
What is Electrorefining explain?
noun. the process for refining a metal in an electrolytic cell, in which the impure metal is used as the anode and the refined metal is deposited on the cathode.
What happens in electrorefining of copper?
Electrorefining Copper. The electrolyte containing the copper ions flows through tank. The electrolyte is often copper dissolved in sulfuric acid which produces an aqueous solution of copper ions. An external power supply is used to pull electrons out of the anode and push them to the cathode.
What is an ore explain the process of electrorefining of copper metal?
In the electrolytic refining of copper, the electrolyte is a solution of acidified copper sulphate. The anode is impure copper, whereas the cathode is a strip of pure copper on passing electric current, pure copper is deposited on the cathode.
Which acts as anode in the electrorefining of metals?
In electrorefining the pure metal acts as cathode and impure metal acts as the anode, whereas the electrolyte is soluble salt of the same metal.
What is meant by refining of metals describe the electrolytic refining of copper with a neat diagram?
In electrolytic refining process, the impure metal is made as anode and a thin strip of pure metal is made as cathode. A solution of the metal salt is made as an electrolyte. On passing the current through the electrolyte, the pure metal from the anode dissolves into the electrolyte.
What is electrorefining explain with example?
Explain with one example. In electrolytic the impure metal is made to act as anode. A strip of the same metal in pure form is used as cathode. They are put in soluble salt of the same metal. The more basic metal remains in solution and less basic go to the anode mud.
What is electrorefining Brainly?
Electrorefining is a process by which metals containing impurities are purified electrolytically to give pure metal.
Why is electrorefining important?
Electrorefining is widely used for the purification and production of copper that is suitable for electrical applications. In an electrorefining process, the anode is the impure metal and the impurities are lost during the passage of metal from the anode to the cathode during electrolysis.
What is the purpose of electrorefining?
The main purpose of electrorefining is to purify the metals. To understand how this process occurs we first know about electrolysis. Electrolysis is the process in which an electric current is passed through a substance to effect a chemical change.
What is electrorefining explain with an example?
Definition of electrorefining : refining of a metal (such as copper) by electrolysis, the crude metal used as the anode going into solution and the pure metal being deposited upon the cathode.
What is electrorefining explain with one example?
Answer: Electrolytic refining is the process of refining impure metals by using electricity. In this process impure metal is made as the anode and a strip of pure metal is made the cathode. A solution of a soluble salt of the same metal is taken as the electrolyte. Example- Copper can be purified by this way.
What is the electrorefining process of copper?
The process for electrorefining copper is typical of those carried out in aqueous solution. In this process, the positive electrode (the anode) is made of the impure copper which is to be purified. The negative electrode (the cathode) is a bar of pure copper.
What is the electrolyte used in electrolytic refining of copper?
In the process of electrolytic refining of copper, the electrolyte used is acidified copper sulphate. Impure metal is made as an anode and pure metal is made as a cathode. On passing the electric current through the electrolyte, the impure metal from anode dissolves into the electrolyte solution
How does electrorefining purify metals?
Electrorefining can be used to purify a number of metals including copper, nickel, cobalt, lead and tin. During the electrorefining stage of copper production, a series of impure copper anodes and thin starter sheet cathodes (such as thin copper foil) are suspended in a tank. The electrolyte containing the copper ions flows through tank.
What is elect electrorefining?
Electrorefining refers to the process of using electrolysis to increase the purity of a metal extracted from its ore (compound or mixture of compounds from which a metal can be extracted commercially). The anode, positive electrode, is the impure metal to be purified.