What are the characteristics of molecular compound?
Physical Properties of Molecular Compounds
| Property | Ionic Compounds | Molecular Compounds |
|---|---|---|
| Physical state at room temperature | Solid | Gas, liquid, or solid |
| Water solubility | Usually high | Variable |
| Melting and boiling temperatures | Generally high | Generally low |
| Electrical conductivity | Good when molten or in solution | Poor |
What are the 3 characteristics of molecular compounds?
Properties of Covalent Molecular Compounds.
- Low melting points and boiling points.
- Low enthalpies of fusion and vaporization These properties are usually one or two orders of magnitude smaller than they are for ionic compounds.
- Soft or brittle solid forms.
- Poor electrical and thermal conductivity.
Which is not a characteristic of a compound?
A compound is a substance formed when two or more elements chemically bonded together. So, all particles of the compound cannot have the same particles.
What is not a molecular compound?
Molecule is the general term used to describe any atoms that are connected by chemical bonds. A compound is a molecule made of atoms from different elements. All compounds are molecules, but not all molecules are compounds. Hydrogen gas (H2) is a molecule, but not a compound because it is made of only one element.
Which of the following is a characteristic of molecular compounds quizlet?
Molecular compounds typically have low melting and boiling points. Molecular compounds are composed of discrete molecules. Under normal conditions molecular compounds often exist as gases, liquids, or low-melting solids.
Which is not true of atoms?
Answer: (a) Atoms are not able to exist independently It is not true that atoms cannot exist independently. Atoms of most elements cannot exist independently, although inert gases can exist as atoms.
Which characteristic describes a compound but not a mixture?
A compound contains atoms of different elements chemically combined together in a fixed ratio. A mixture is a combination of two or more substances where there is no chemical combination or reaction. Compounds contain different elements in a fixed ratio arranged in a defined manner through chemical bonds.
What are non examples of molecules?
Any of the noble gasses: He, Ne, Ar, Kr, Xe (These are atoms, not technically molecules.) Any of the homonuclear diatomic elements: H2, N2, O2, Cl2 (These are truly nonpolar molecules.) Carbon dioxide – CO. Benzene – C6H.
What substances are not compounds?
The following are examples of NOT compounds.
- Br2 , bromine, is a diatomic element, or a molecular element.
- Ar , argon, is an atom; thus, it cannot be multi-atomic, and consequently is not a compound.
- S8 , sulfur, is an octatomic element, or a molecular element.
Which is not characteristic of a covalent compound?
They have no definite geometry: covalent bonds are directional in nature and as a result they have definite geometry and shape. Hence, this statement is wrong.
What are the characteristics of molecular compounds?
Under normal conditions, molecular compounds often exist as gases, low-boiling liquids, and low-melting solids, although many important exceptions exist. Whereas ionic compounds are usually formed when a metal and a nonmetal combine, covalent compounds are usually formed by a combination of nonmetals.
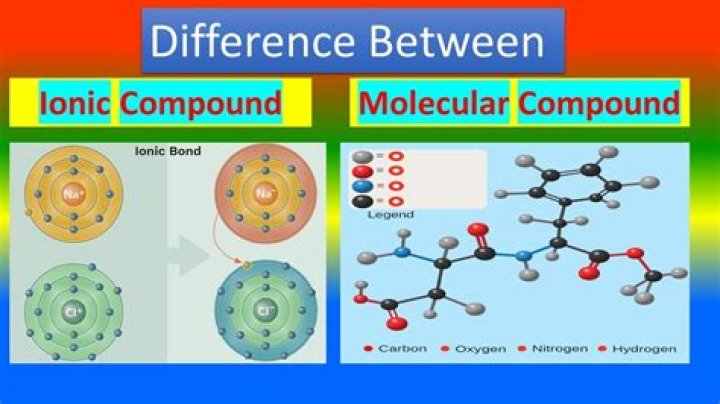
What is the difference between an ionic compound and molecular compound?
The main difference between an ionic compound and a molecular compound is the constituent elements. In an ionic compound, metal element and nonmetal element combine together to form compounds.
What is the difference between polar and non-polar molecules?
If the combining elements have a separate charge specific to each of them, then the molecular compound is referred to as polar molecule. On the contrary, non-polar molecules are those, which constituent elements have no separate charges. The main difference between an ionic compound and a molecular compound is the constituent elements.
Which is not a characteristic of a covalent compound?
No definite geometry is not a characteristic of a covalent compound. The covalent compound has definite geometry because of the bond forms. Was this answer helpful?