Is iron an element compound or mixture?
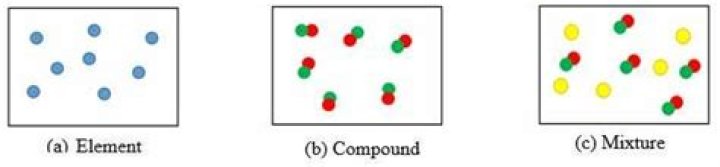
Element: An element cannot be broken down into simpler substances. Compound: A compound is composed of two or more elements chemically combined….Chemistry: 12. Elements, Compounds and Mixtures.
| Compound | Iron sulphide |
|---|---|
| Symbol | FeS |
| State of matter (at room temp) | solid |
| Elements in the compound (and state at room temp) | Iron (solid) Sulphur (solid) |
Is iron an element or compound Why?
Iron is a chemical element with symbol Fe and atomic number 26. Classified as a transition metal, Iron is a solid at room temperature.
Is iron Fe mixture?
Natural iron is a mixture of four stable isotopes: iron-56 (91.66 percent), iron-54 (5.82 percent), iron-57 (2.19 percent), and iron-58 (0.33 percent).
Is iron a homogeneous mixture?
Iron ore is heterogeneous. A heterogeneous substance is one that contains two or more different substances that are NOT chemically combined together,…
Is iron An example of a compound?
Iron is one of the most common elements found on the earth. It can form several different compounds due to the fact that it is commonly found in both the +2 (ferrous) and +3 (ferric) state.
Is iron a mineral or metal?
Iron (/ˈaɪərn/) is a chemical element with symbol Fe (from Latin: ferrum) and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table.
What is iron mixture?
A mixture is made from different substances that are not chemically joined. For example, powdered iron and powdered sulfur mixed together make a mixture of iron and sulfur. There are times when the purity of a substance is very important. For example, a medicine must not contain any harmful chemicals.
Is iron ore a mixture or pure substance?
How is the element iron formed?
Iron is made inside stars, specifically red super-giants. The elements form together inside a star during fusion. When the supernova occurs, the iron fragments are blasted into the space. This is how Iron came to Earth millions of years ago.
How is an iron formed?
Today, iron is made by heating hematite or magnetite in a blast furnace along with with a form of carbon called “coke” as well as calcium carbonate (CaCO3), better known as limestone.
What is iron compound?
Three oxygen compounds of iron are known: ferrous oxide, FeO; ferric oxide, Fe2O3; and ferrosoferric oxide, or ferroferric oxide, Fe3O4, which contains iron in both +2 and +3 oxidation states. Ferrous oxide is a greenish to black powder used primarily as a pigment for glasses.
What are the common compounds of iron?
Hydrides. The term hydride is used to indicate compounds of the type MxHyand not necessarily to indicate that any compounds listed behave as hydrides chemically.
What are common uses of iron?
Iron catalysts are used in the Haber-Bosch Process to produce ammonia and the Fischer-Tropsch Process to convert carbon monoxide to hydrocarbons used for fuels and lubricants.
What is the difference between a compound and a mixture?
The compound is used to mean a substance,created by combining two or more than two substances chemically in a certain ratio by weight.
Is iron a pure substance?
A pure substance refers to an element or a compound that has no component of another compound or element. Pure substances are made of only one type of atom or molecule. Hydrogen gas and pure iron are examples of pure substances. Hydrogen consists of hydrogen atoms only while iron consists of only iron atoms.