Is H3AsO4 oxidizing or reducing agent?
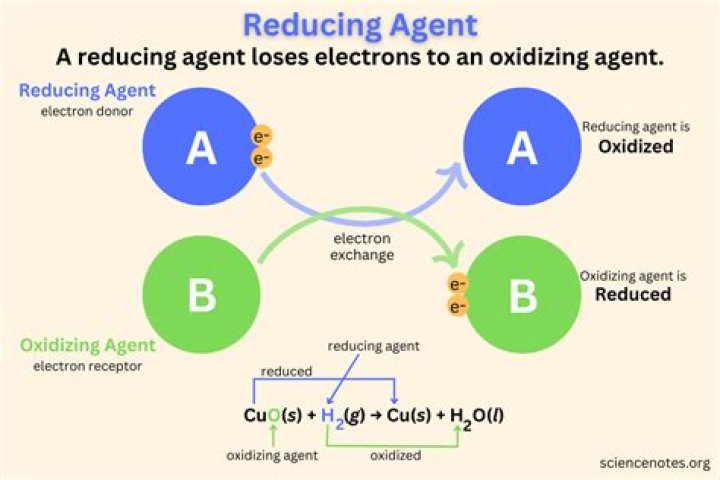
H 3AsO 4 is an oxidizing agent, H 2SO 3 is a reducing agent.
What is the correct name for H3AsO4?
Arsenic acid
Arsenic acid
| PubChem CID | 234 |
|---|---|
| Structure | Find Similar Structures |
| Chemical Safety | Laboratory Chemical Safety Summary (LCSS) Datasheet |
| Molecular Formula | AsH3O4 or H3AsO4 |
| Synonyms | Arsenic acid Orthoarsenic acid arsoric acid Arsenic acid (H3AsO4) 7778-39-4 More… |
What is the chemical name of arsenic?
symbol As
Arsenic is a chemical element with the symbol As and atomic number 33. Arsenic occurs in many minerals, usually in combination with sulfur and metals, but also as a pure elemental crystal. Arsenic is a metalloid.
How do you find the element oxidized and the element reduced?
Oxidation numbers represent the potential charge of an atom in its ionic state. If an atom’s oxidation number decreases in a reaction, it is reduced. If an atom’s oxidation number increases, it is oxidized.
Is ch4 a reducing agent?
In the combustion of methane molecular oxygen is the oxidizing agent and methane is the reducing agent. In the catalytic hydrogenation of propene, molecular hydrogen is the reducing agent and propene is the oxidizing agent.
What is the conjugate base of H3AsO4?
Arsenate
Arsenic acid
| Names | |
|---|---|
| Conjugate base | Arsenate |
| Structure | |
| Molecular shape | Tetrahedral |
| Hazards |
How many arsenic atoms are in H3AsO4?
H3AsO4 is 3 atoms of hydrogen, 1 atom of arsenic, and 4 atoms of oxygen.
What is the oxidation number of arsenic?
arsenic
| atomic number | 33 |
|---|---|
| density | |
| (gray form) | 5.73 g/cm3 at 14 °C (57 °F) |
| (yellow form) | 2.03 g/cm3 at 18 °C (64 °F) |
| oxidation states | -3, +3, +5 |
Which element is oxidized and which is reduced in 3fe no3 2 aq )+ 2al s → 3fe s )+ 2al no3 3 AQ?
Aluminum goes from an oxidation state of 0 to 3+. Therefore, it has lost electrons and is oxidized.