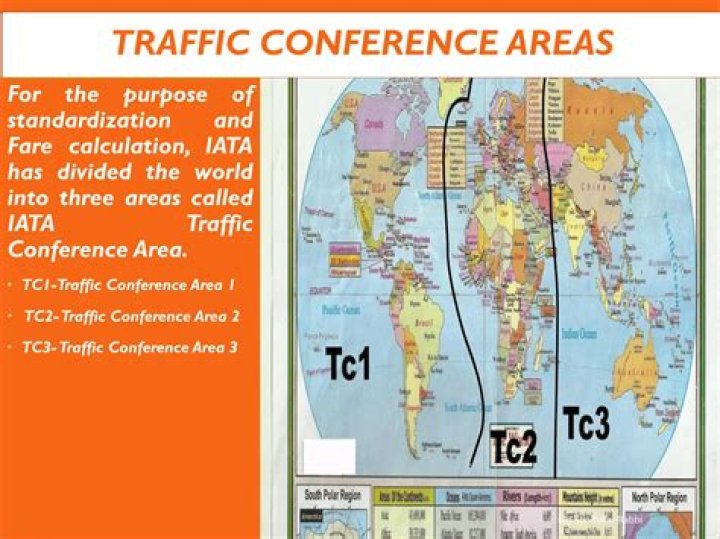
Is dolomite a primary or secondary mineral?

Occurrence and Compositon Calcite and dolomite are common as primary minerals in a wide variety of sedimentary and metamorphic rocks. The may also form as secondary minerals. Calcite is CaCO3; dolomite is CaMg(CO3)2.
What type of minerals are calcite and dolomite?
Limestones and dolomites are collectively referred to as carbonates because they consist predominantly of the carbonate minerals calcite (CaCO3) and dolomite (CaMg[CO3]2).
What group do calcite and dolomite belong to?
carbonates
The carbonates also include the dolomite group (Table 3.7). Table 3.7. Minerals of calcite and aragonite groups. The important petrogenic elements/minerals from carbonate group are calcium, magnesium, and iron carbonates or aragonite, calcite and dolomite, less-frequent siderite, and very rare magnesite.
What type of mineral is rhodochrosite?
manganese carbonate
Rhodochrosite is a manganese carbonate (MnCO3) mineral. The color is brilliant and transparent yellow, pink, and rose/cherry-red (Fig. 1.45) with 47.79% Mn in its purest form.
Is calcite a mineral?
Calcite: A mineral consisting largely of calcium carbonate (CaCO3 ). Next to quartz, it is the most abundant of the Earth’s minerals. Crystallizing in the hexagonal system, calcite is noted for its wide variety of crystalline forms.
What is primary dolomite?
Primary vs Secondary Dolomite: As suggested by Zenger (1972), Land (1980), and others, primary dolomite is used to refer to dolomites formed by direct nucle- ation of the crystals in the water column. Secondary dolomites are those formed during diagenesis via replacement or cementation of existing carbonate.
What do calcite and dolomite have in common?
Calcite and dolomite are very similar minerals. Both have the same hardness (H = 3), the same rhombohedral cleavage, and are found in identical geologic settings.
What are the examples of calcite?
Geological sources. Calcite, aragonite and vaterite are pure calcium carbonate minerals. Industrially important source rocks which are predominantly calcium carbonate include limestone, chalk, marble and travertine.
How do calcite and dolomite differ?
Calcite mainly contains calcium carbonate and dolomite contains calcium magnesium carbonate. Dolomite differs from calcite because of the presence of magnesium. Calcite reacts quickly with acids and produce carbon dioxide bubbles. But dolomite is weakly reacts with acids producing bubbles very slowly.
Is rhodochrosite a calcite?
Rhodochrosite belongs to the calcite group of minerals, a group of related carbonates that are isomorphous with one another. They are similar in many physical properties, and may partially or fully replace one another, forming a solid solution series.
How many types of calcite are there?
More than 300 forms of calcite have been recognized.
What is the decomposition of dolomite and calcite?
Calcite decomposes to form lime (CaO) at around 900°C, and dolomite decomposes in a two-stage process to form both lime and periclase (MgO). Calcite, crystallizing in the rhombohedral crystal system is the thermodynamically stable phase of calcium carbonate.
What is an example of a secondary mineral?
Secondary minerals may also form by oxidations reactions when primary minerals react with oxygen in air or water. For example, oxidation of iron-rich olivine or pyroxene commonly produces hematite (Fe 2 O 3 ). Mineral matter remaining after chemical weathering often includes original mineral grains that did not decompose.
Where does ridrhodochrosite come from?
Rhodochrosite occurs in bedded, marine, sedimentary deposits along with various Mn oxides.
What is the solubility of calcite?
At earth surface temperatures and pressures, therefore, common carbonate mineral species exhibit a hierarchy of solubility, with magnesian calcite the most soluble. Aragonite, calcite and ideal dolomite become progressively less soluble, or more stable. Protodolomite may actually be less soluble than calcite.