Is an amide an amine?
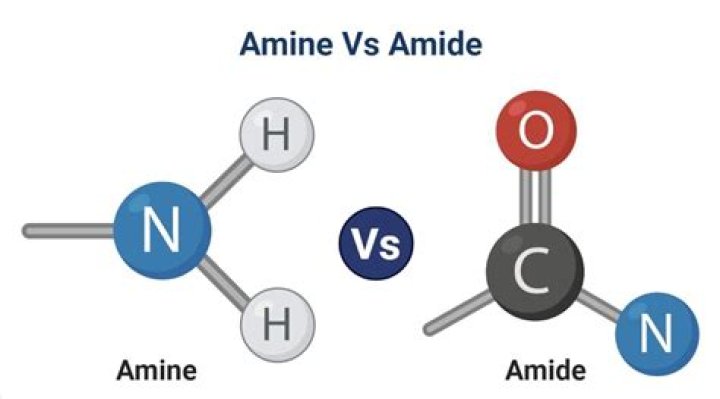
Amines and amides are two types of compounds found in the field of organic chemistry. The main difference between amine and amide is the presence of a carbonyl group in their structure; amines have no carbonyl groups attached to the nitrogen atom whereas amides have a carbonyl group attached to a nitrogen atom.
Which group is the amine group?
Inorganic derivatives of ammonia are also called amines, such as monochloramine (NClH2). The substituent -NH2 is called an amino group. Compounds with a nitrogen atom attached to a carbonyl group, thus having the structure R–CO–NR′R″, are called amides and have different chemical properties from amines.
What functional group is an amide?
carbonyl functional group
Amides are compounds that consist of a carbonyl functional group which is connected to both an amine group and a hydrocarbon group (or hydrogen atom). A carbonyl functional group consists of a carbon atom which is double-bonded with an oxygen atom.
Does amide have a carbonyl group?
In amides, the carbonyl carbon is bonded to a nitrogen. The nitrogen in an amide can be bonded either to hydrogens, to carbons, or to both. Another way of thinking of an amide is that it is a carbonyl bonded to an amine. In esters, the carbonyl carbon is bonded to an oxygen which is itself bonded to another carbon.
Does amide contain a carbonyl group?
The amide. functional group has a carbonyl group joined to a nitrogen atom from ammonia or an amine. The properties of the amide functional group differ from those of the simple carbonyl group, NH 3, and amines.
Is amine a functional group?
amine: Organic compounds or the functional group that contains a basic nitrogen atom with a lone pair.
What do you mean by amide?
1 : an inorganic compound derived from ammonia by replacement of an atom of hydrogen with another element (such as a metal) 2 : any of a class of organic compounds derived from ammonia or an amine by replacement of hydrogen with an acyl group — compare amine, imide.
What is an amide group?
Amide, an organic functional group characterized by a carbonyl group linked to a nitrogen atom, or a compound that contains this functional group. (CO=NH2) Peptide bond, a chemical bond formed between two molecules when the carboxyl group of one molecule reacts with the amine group of the other molecule.
What is the formula for an amine group?
Chemically speaking, an amino acid is a carboxylic acid which has an amine group attached to it. The general linear formula of an amino acid is R-CH(NH2)-COOH.
Amide, an organic functional group characterized by a carbonyl group linked to a nitrogen atom, or a compound that contains this functional group.
What is the functional group of amide?
Amide group may refer to: Amide, an organic functional group characterized by a carbonyl group linked to a nitrogen atom , or a compound that contains this functional group. Peptide bond, a chemical bond formed between two molecules when the carboxyl group of one molecule reacts with the amine group of the other molecule.
Which compound is an amide?
An amide (/ˈæmaɪd/ or /ˈæmɪd/ or /ˈeɪmaɪd/), also known as an acid amide, is a compound with the functional group RnE(O)xNR′2 (R and R′ refer to H or organic groups).



