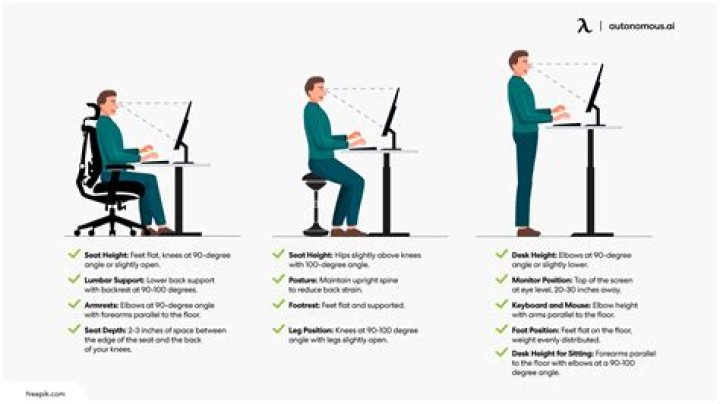
Is Aldopentose a sugar?
Arabinose is an aldopentose, a simple five-carbon sugar. In nature, l-arabinose rarely exists in the form of simple sugar, it was combined with other monosaccharides usually.
Which sugar is an example for a Aldopentose?
D-ribose
The most abundant five-carbon sugars are L-arabinose, D-ribose, 2-deoxy-D-ribose,1 and D-xylose, which all are aldopentoses.
What is sugar cyclization?
Cyclization of Monosaccharides When an aldose cyclizes, the hydroxyl group on the second to last carbon undergoes an intramolecular reaction with the carbonyl group of the aldehyde. The product resulting from aldose cyclization is a hemiacetal. The product resulting from ketose cyclization is a hemiketal.
What are Aldohexose sugars?
Glucose (also known as dextrose) is a carbohydrate compound consisting of six carbon atoms and an aldehyde group and they are referred to as aldohexose. It is a carbohydrate and is the most important simple sugar (monosaccharide) in animal metabolism.
What is an Aldopentose sugar?
aldopentose. Definition : A pentose with a (potential) aldehyde group at one end. pentose. Definition : A five-carbon monosaccharide which in its linear form contains either an aldehyde group at position 1 (aldopentose) or a ketone group at position 2 (ketopentose).
Is Sucrose a white sugar?
Sucrose is crystallised white sugar produced by the sugar cane plant and can be found in households and foods worldwide. Sucrose is a disaccharide made up of 50% glucose and 50% fructose and is broken down rapidly into its constituent parts. Due to its glucose content, sucrose has a GI value of 65.
Which of following is an example of an Aldopentose?
D-Ribose
D-Ribose → Example of aldopentose.
Is D Ribose an example of Aldopentose?
Ribose is a sugar that is obtained by the hydrolysis of the RNA molecule. It has five carbon atoms in the ring. It contains aldehyde groups. Hence, it is aldopentose.
What does the cyclization of glucose do?
Cyclization of glucose to its hemiacetal form Glucose has an aldehyde group and five hydroxyl groups. Does that ring a bell? Yes, glucose can form an intramolecular cyclic hemiacetal. Let’s now show the formation of hemiacetal of glucose starting from its open structure (Fischer projection).
What are Hemiacetals and Hemiketals?
When an alcohol adds to an aldehyde, the result is called a hemiacetal; when an alcohol adds to a ketone the resulting product is a hemiketal. A hydrate forms as the result of a water molecule adding to the carbonyl carbon of the aldehyde or ketone.