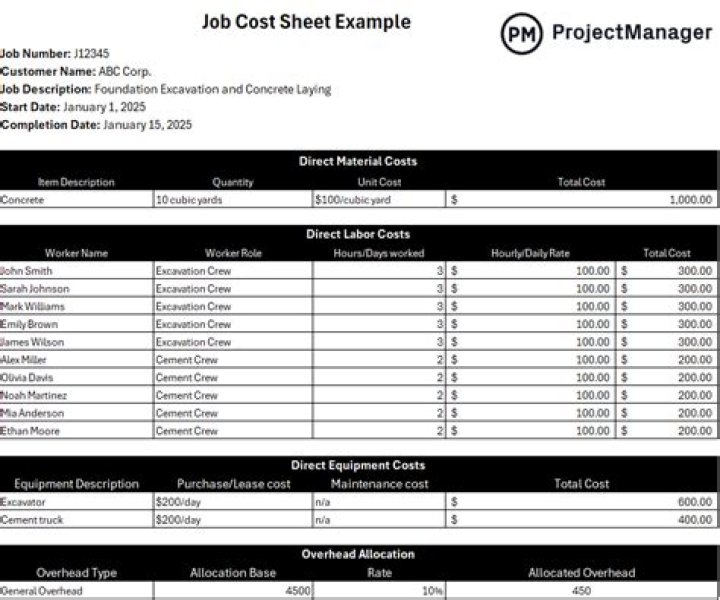
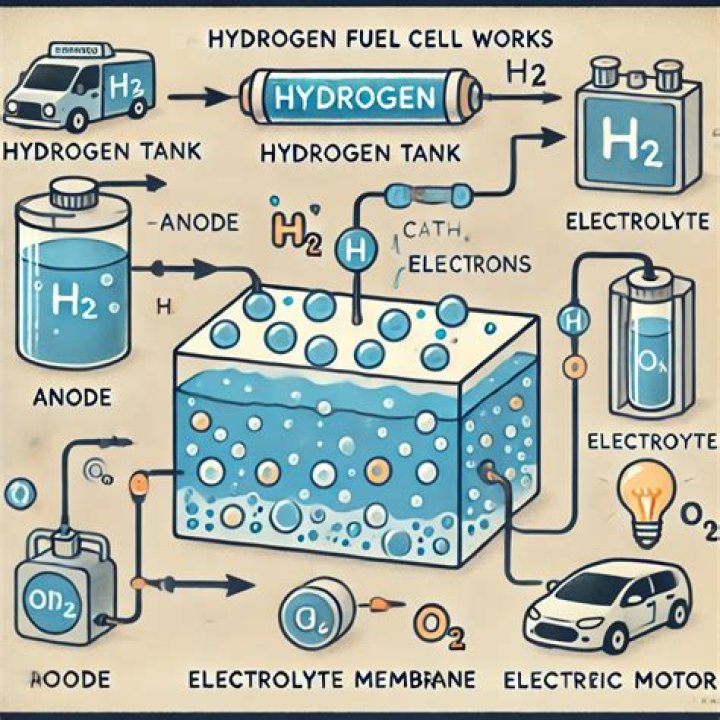
How is hydrogen produced for fuel cells?
Hydrogen fuel can be produced from methane or by electrolysis of water. As of 2020, the majority of hydrogen (∼95%) is produced from fossil fuels by steam reforming or partial oxidation of methane and coal gasification with only a small quantity by other routes such as biomass gasification or electrolysis of water.
What is fuel cell h2 o2 fuel cell?
In a fuel cell, hydrogen and oxygen are combined to generate electricity, heat, and water. The protons pass through the porous electrolyte membrane, while the electrons are forced through a circuit, generating an electric current and excess heat.
Is hydrogen toxic or nontoxic?
For example, hydrogen is non-toxic. Specifically, hydrogen has a wide range of flammable concentrations in air and lower ignition energy than gasoline or natural gas, which means it can ignite more easily.
How do alkaline fuel cells work?
Alkaline fuel cells consume hydrogen and pure oxygen, to produce potable water, heat, and electricity. They are among the most efficient fuel cells, having the potential to reach 70%. NASA has used alkaline fuel cells since the mid-1960s, in the Apollo-series missions and on the Space Shuttle.
What are the two most common ways to produce hydrogen gas used in fuel cells?
The two most common methods for producing hydrogen are steam-methane reforming and electrolysis (splitting water with electricity.
How does hydrogen become oxygen?
Electrolysis is the process of using electricity to split water into hydrogen and oxygen. This reaction takes place in a unit called an electrolyzer.
Where are alkaline fuel cells used?
Alkaline fuel cells (AFCs) are one of the most developed technologies and have been used since the mid-1960s by NASA in the Apollo and space shuttle programs. The fuel cells on board these spacecraft provide electrical power for onboard systems, as well as drinking water.