Does a catalyst affect the activation energy of the reverse reaction?
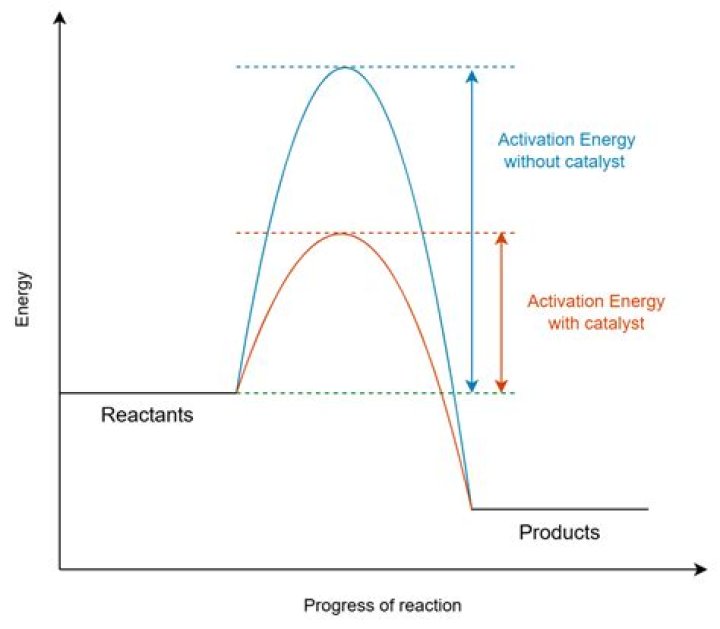
Catalysts lower the activation energy for the reaction. Catalysts will not alter the equilibrium of the reaction. Catalysts increase the forward rate, while reducing the reverse rate.
How does catalyst affect reverse reaction?
In the presence of a catalyst, both the forward and reverse reaction rates will speed up equally, thereby allowing the system to reach equilibrium faster. In the presence of a catalyst, the same amounts of reactants and products will be present at equilibrium as there would be in the uncatalyzed reaction.
What is the effect of a catalyst on the activation energy?
A catalyst speeds up the rate of a reaction by lowering the activation energy; in addition, the catalyst is regenerated in the process.
What is the effect of a catalyst on the activation energy of a reaction on the δg0?
Catalysts impact the rate of the reaction which is a kinetic function. It lowers the activation energy of the reaction, and therefore, it shorten the pathway of the reaction without changing the energies of the reactants or products, and therefore, no change on the free energy.
How does a catalyst decrease activation energy?
The catalyst lowers the energy of the transition state for the reaction. Since the activation energy is the difference between the transition state energy and the reactant energy, lowering the transition state energy also lowers the activation energy.
What is the effect of catalyst on the rate of reaction increase or decrease?
The rate of a reaction can be increased by adding a suitable catalyst. A catalyst is a substance which increases the rate of a chemical reaction but it is not used up (remains chemically unchanged at the end). It provides an alternative reaction pathway of lower activation energy.
Which statement about the effect of a catalyst on a reversible reaction is correct?
On the velocity in a reversible reaction, the correct explanation of the effect of catalyst is. Effect of catalyst is that it attains equilibrium quickly by providing a new reaction path of low activation energy. It does not alter the state of equilibrium.
What is the effect of catalyst on Gibbs energy change in activation energy?
The catalyst does not affect enthalpy, entropy and temperature remain the same therefore there is no effect on Gibbs free energy .
Does a catalyst lower activation energy?
A catalyst provides an alternative route for the reaction with a lower activation energy. It does not “lower the activation energy of the reaction”.
How does a catalyst lower the activation energy?
A catalyst works by providing a different pathway for the reaction, one that has a lower activation energy than the uncatalyzed pathway. This lower activation energy means that a larger fraction of collisions are successful at a given temperature, leading to an increased reaction rate.
Why do Catalysts increase the rate of reaction?
A catalyst increases the rate of reaction in a slightly unconventional way from other means of increasing reaction rate. The role of a catalyst is to lower the activation energy so that a greater proportion of the particles have enough energy to react.
When a catalyst is added to a reversible reaction in equilibrium state the value of the equilibrium constant?
NEET Question. Catalyst does not alter the Position of Equilibrium but it helps to attain equilibrium position quickly therefore equilibrium constant value doesn’t change as the position of equilibrium remains same.
How does adding a catalyst affect the activation energy?
Adding a catalyst has this effect on activation energy. A catalyst provides an alternative route for the reaction with a lower activation energy. This is illustrated on the following energy profile: A word of caution
Why do catalysts increase the forward and reverse reactions the same?
With reference to the picture below, how is it that when there is a lower activation energy (due to the alternate reaction pathway provided by the catalyst) that the catalyst will increase the forward and reverse reactions exactly the same amount.
How do you change the activation energy of a chemical reaction?
One possible way of doing this is to provide an alternative way for the reaction to happen which has a lower activation energy. In other words, to move the activation energy to the left on the graph: Adding a catalyst has this effect on activation energy. A catalyst provides an alternative route for the reaction with a lower activation energy.
How to move the activation energy to the left?
In other words, to move the activation energy to the left on the graph: Adding a catalyst has this effect on activation energy. A catalyst provides an alternative route for the reaction with a lower activation energy. This is illustrated on the following energy profile: