Do electrons flow in the salt bridge?
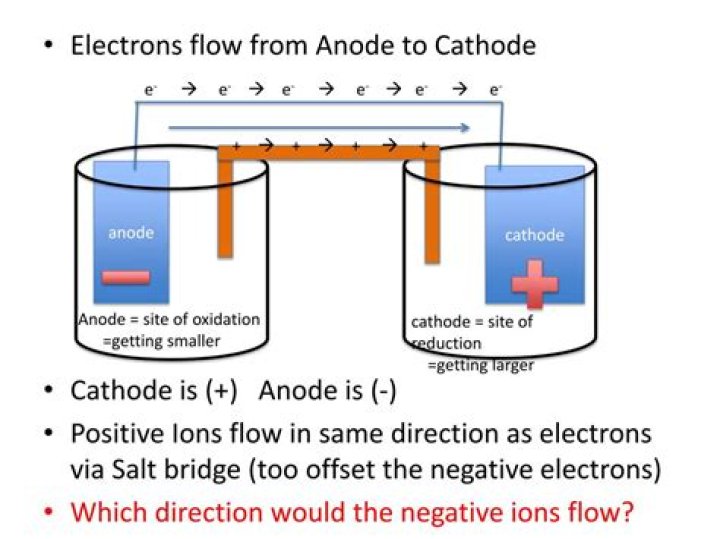
Electrons won’t move through the salt bridge, and won’t go back through the wire (because of the unfavorable redox conditions). Instead, ions from the solution move. Cl- ions from the negative cell B move to cell A to try to “equalize” the charge, which allows for electricity to continue flowing.
What is the purpose of a salt bridge in an electrochemical cell?
The purpose of the salt bridge is to act as a source of spectator ions that can migrate into each of the half cells to preserve neutrality. Any charge buildup in the solutions of the two half cells is known as a junction potential.
What flows through the salt bridge?
Reduction describes the gain of electrons by a molecule, atom, or ion. The electrons always flow from the anode to the cathode. The half-cells are connected by a salt bridge that allows the ions in the solution to move from one half-cell to the other, so that the reaction can continue.
Which way do electrons flow in electrolysis?
anode
Electrons flow from anode to cathode (this is always the case). For an electrolytic cell however, this flow is not spontaneous but must be driven by an external power source.
Which way do ions flow in a salt bridge?
Anions in the salt bridge flow toward the anode and cations in the salt bridge flow toward the cathode. The movement of these ions completes the circuit and keeps each half-cell electrically neutral.
Do electrons flow through the wire?
Electrons do not move along a wire like cars on a highway. Each atom has electrons in it. If you put new electrons in a conductor, they will join atoms, and each atom will deliver an electron to the next atom. This next atom takes in the electron and sends out another one on the other side.
How does salt bridge maintain electrical neutrality?
ions in the half cell. This prevents the flow of electrons to the copper plate. Similarly, it supplies positive ions to the reduction half-cell in order to neutralise the excess of sulphate ions. Thus, salt bridge maintains the electrical neutrality in both the half cells.
What is the main function of salt bridge?
The main function of a salt bridge is to maintain the electrical neutrality of both electrolytic solutions within the internal circuit. It basically helps in preventing the accumulation of positive and negative charges around the respective electrolytic electrodes and further allowing a smooth reaction to occur.
What is conventional electron flow?
Conventional Current assumes that current flows out of the positive terminal, through the circuit and into the negative terminal of the source. Electron Flow is what actually happens and electrons flow out of the negative terminal, through the circuit and into the positive terminal of the source.
Do electrons always flow from anode to cathode?
Electrons always flow from the anode to the cathode or from the oxidation half cell to the reduction half cell. In terms of Eocell of the half reactions, the electrons will flow from the more negative half reaction to the more positive half reaction. A cell diagram is a representation of an electrochemical cell.
Do ions pass through salt bridge?
Ions leave the salt bridge and move further, without accumulating at the bridge end. For all salt bridges where there is no other reactions involved, so all simple salts in solution or molten salts. Ions move all the way through. There is some concentration difference in salt bridge and main half cell volume.
Do anions go through the salt bridge?
Anions flow towards the anode through the salt bridge. They flow in the opposite direction of the electrons.